
Structure of prodrug-transporter complex revealed
Scientists develop structural model that could help in the development of drugs with increased absorption rates
Prodrugs, inactive medications that are metabolised into an active form within the body, are able to hijack human peptide transporters PepT1 or PepT2 and hitch a ride directly into a cell. This process allows for the accelerated absorption of a drug into a patient’s bloodstream. While prodrugs are clinically effective, little is known about the peptide transporters themselves at the molecular level.
Research groups lead by CSSB/EMBL scientists Christian Löw and Jan Kosinski are now a step closer to understanding the structures of peptide transporters and how they recognise, bind and transport prodrugs. The groups recently determined a high-resolution crystal structure of a peptide transporter in complex with the prodrug valganciclovir: a medication that combats certain viral infections. Their results, published in the Journal of the American Chemical Society, could assist in the design of prodrugs with improved absorption rates.
Bacterial relations
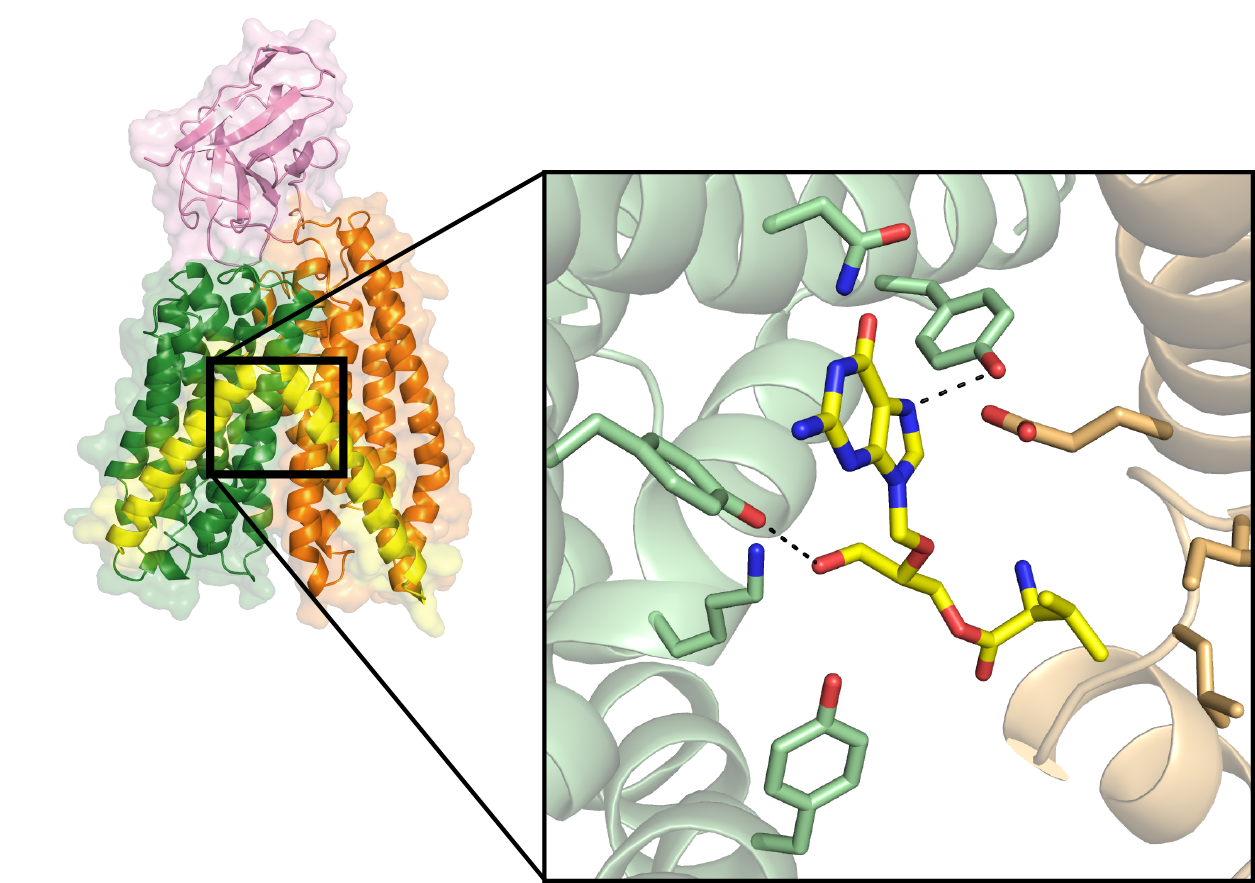
The scientists chose to study the bacterial peptide transporter DtpA, from E. coli, because of its close evolutional relationship with the human PepT1 transporter. To produce well-diffracting crystals of DtpA, Löw teamed up with the group of Jan Steyaert from Belgium to generate DtpA-specific nanobodies. Nanobodies are antibody fragments derived from naturally-occurring, heavy-chain-only antibodies found in llamas. “The nanobodies act as crystallisation chaperones,” explains Löw. “They increase the stability of membrane proteins such as DtpA, thus allowing us to generate high-quality crystals.”
Yonca Ural-Blimke, of the Löw group, then carried out the crystallisation experiments on DtpA in complex with a nanobody and a prodrug molecule at the Sample Preparation and Characterization facility (SPC). She analysed the obtained crystals at the EMBL-operated beamlines P13 and P14: part of DESY’s X-ray light source PETRA III. The determined 2.65 Ångstrom-resolution structure of DtpA bound with the prodrug valganciclovir revealed an unexpected result. “The orientation of the prodrug in the binding pocket is flipped by 180 degrees relative to prior predictions,” says Löw. “We also discovered that DtpA preferably binds, and likely transports, tripeptides over dipeptides.”
The Kosinski group built a high-quality structural model of the human PepT1 transporter in complex with valganciclovir using the DtpA crystal structure as a guide. “The model,” Kosinski explains, “shows that human PepT1 very likely binds the drug in the same way as DtpA and pinpoints the exact amino acid groups responsible for binding.” This research could facilitate the development of new prodrugs with improved absorption rates that could lower pharmacologically effective doses and reduce the negative side effects experienced by patients.
Adapted from the news article on the CSSB website


