
Crystals in the beamlight
New system developed at EMBL automates crystals preparation for X-ray analysis
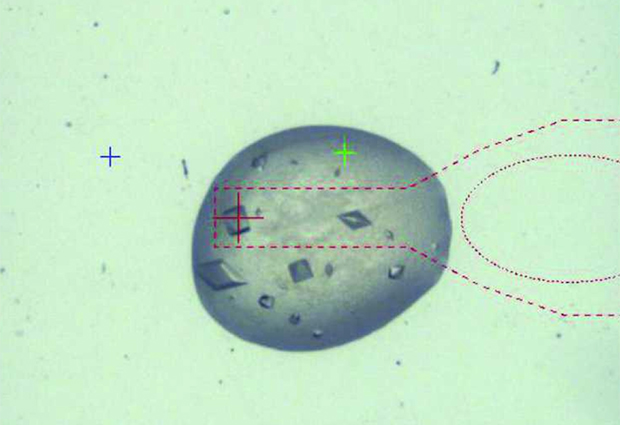
CrystalDirect streamlines the process of preparing crystals for X-ray diffraction experiments. Whereas scientists currently have to harvest and treat the crystals by hand, samples processed at EMBL’s high-throughput crystallisation (HTX) lab in Grenoble can now be loaded in the system at one end and the relevant data collected at the other, with little more than a mouse-click in-between.
This fully automated system, described in a recent article in Acta Crystallographica Section D, is making crystallography much more accessible, also for scientists from other fields who may not have the training required to handle fragile crystals. It could pave the way for a much wider use of the technique and for collaborations with other scientific fields.
Following crystallisation, a typical crystal has to go through several steps: harvesting, soaking, and cryo-cooling, before it can be analysed by X-rays. Until now these had to be done by hand and required a lot of time and skills. The teams of Jose Antonio Marquez and Florent Cipriani, both at EMBL-Grenoble, collaborated to automate this process with some creative innovations.
Crystals are now grown on a very thin plastic film that can be laser-cut and carried automatically to the next processing step, thus removing the need to find and harvest the microscopic samples by hand. It makes the process much smoother and safer for the crystals that undergo less mechanical stress.
When applied to structure-based drug design projects, in which samples have to be soaked with a ligand before X-ray analysis, the plastic film becomes a gentle mixer. The solution with the ligand is deposited on top of the film, above the crystal, and a robot punctures a tiny hole in it to allow the ligand to mix slowly and gently with the crystal and bind together. This process is much less damaging to the crystal than the traditional hand-harvesting and soaking, thus allowing for more crystals to be prepared and more ligands to be tested.
Finally, most protein crystals have to be cooled to very low temperatures before X-ray analysis. At that stage the plastic film becomes a sieve when the robot punctures another tiny hole in it to gently remove all the solution surrounding the crystal. The sample can then be dipped in liquid nitrogen and cryo-cooled directly and in isolation. Traditionally this step was preceded by the addition of chemicals, like glycerol, to protect the crystals from the damage caused by the freezing of water in the solution; without water around this step is not necessary, thus removing the risk of chemical damage to the crystal.
These developments improve the quality and the quantity of the crystals that finally reach the X-rays for analysis. They also make crystallography easier to access since scientists can control everything remotely through the web-based crystallization information management system (CRIMS), potentially opening up the field to many more applications.
Access to this new method for processing crystals has been provided by the HTX lab in Grenoble for over a year; the EC-funded iNEXT project will provide funded access to scientists outside of EMBL until 2019.


