
Translating blue-sky research into the clinic
What does it take to create a vaccine or improve cancer therapies?
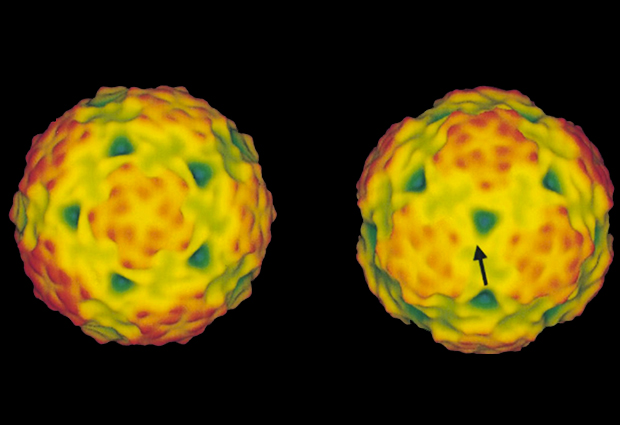
In the summer of 1999, Ilaria Ferlenghi sat at a microscope, staring intensely at the greyscale image swimming into and out of focus on screen. She was inside the microscopy facility at EMBL’s Heidelberg site, using one of two cryo-electron microscopes. Within the blur, she was searching for a glimpse of the tick-borne encephalitis virus (TBEV) – a virus for which south-west Germany is particularly renowned. In the forest outside the laboratory, hikers were attempting to avoid TBEV. Those sensible enough covered their ankles, to protect themselves from TBEV-infected ticks lurking in the undergrowth. Anyone infected with TBEV risks fever, seizures and even death.
Discovering the viral gatekeepers
TBEV’s symptoms in humans arise because the virus invades and disrupts the central nervous system. After a bite from an infected tick, proteins that form the outer shell of TBEV enable the virus to invade cells. Some of these proteins act to protect the virus’s genetic material from harm, while others interact specifically with proteins on the surface of the host cell. Like a key in a lock, this interaction between viral and cell proteins grants the virus access to the cell. There, it can hijack the molecular machinery needed to replicate itself many times over, damaging cells and causing clinical symptoms in the process.
Part of Ferlenghi’s research at EMBL involved imaging the whole virus as well as discovering the structure of the surrounding surface proteins, mainly using cryo-electron microscopy (cryo-EM). At the time, however, cryo-EM wasn’t considered a particularly important technique for revealing protein structures. Other research groups were using X-ray crystallography instead, and in doing so could achieve much higher-resolution images.

Yet the use of just one imaging technique is never enough to be confident about a protein structure – validation via different tools is vital. This was why Ferlenghi wanted to further develop cryo-EM as a structural biology technique by studying TBEV and other viruses. Development of this technique – led by Ferlenghi’s supervisor, the late Steve Fuller – continued the influential work kickstarted by Nobel Prize winner Jacques Dubochet at EMBL in the 1980s.
While at EMBL, Ferlenghi succeeded in her aim. By imaging the structure of the TBEV proteins in both their wild-type and mutant forms, Ferlenghi gained a better understanding of how they interact with proteins on the host cell’s surface. The mechanism by which they enter the cell became clearer too. “At the time we didn’t know if it would be important or not. We were playing with science,” recalls Ferlenghi. “We didn’t set out to save lives.”
Combining chemotherapies
Fast-forward 20 years and EMBL scientists still ‘play with science’. At EMBL, researchers have the freedom to ask fundamental scientific questions that help to further human knowledge. Often, a clinical application might not be foreseeable, but potential applications can strike at any time.
“We were playing with science, we didn’t set out to save lives”
Current research in Christoph Merten’s group, for instance, focuses predominantly on the development and production of microfluidic devices. These widgets combine silicon chips, maze-like plastic tube networks and bespoke computer code to allow automated, high-throughput screening of biological samples. By Merten’s own admission, the instruments – although functional and useful – are often “ugly and not particularly user-friendly”. But, by collaborating with clinicians and other researchers, the group can use the instruments to provide a good proof of principle for clinical applications.

Merten’s latest partnership, with clinicians and researchers at Uniklinik RWTH Aachen and Heidelberg University, enables these microfluidic devices to be used to test the efficacy of chemotherapy combinations on biopsies from cancer patients. When validated in mice, the combined therapies recommended by the microfluidics data were more effective than standard care.
This kind of research is driving the advancement of personalised medicine. But Merten is quick to warn that, although everyone involved in the project is keen to see this knowledge translated into new clinical therapies, there are still many years before that can become a reality. “We would need to be exceptionally confident in our results before we can recommend a treatment to a patient,” says Merten. “People’s lives are at stake.”

Travelling through time and space
It’s sensible for Merten to be cautious. Clinical applications of fundamental research require a lot of investigation, investment by industry, and – importantly – time. For Ferlenghi, this combination has been a recipe for success. Almost 20 years after leaving EMBL, and with several senior research positions behind her, she is now Head of Structural Vaccinology at GlaxoSmithKline (GSK) in Siena, Italy. Alongside her team, she still uses cryo-EM – surprisingly, even the microscope is the same. “After a brief stint with Steve at the University of Oxford, the cryo-EM I used at EMBL is with me again,” says Ferlenghi. “We use it at GSK all the time and it still works!” Now, however, it’s used to look at the protein structures of potential vaccines to help inform their clinical development. One of the most successful candidates to date? A vaccine against TBEV.
This vaccine is given to people throughout the world – including the hikers exploring the forest outside EMBL. Ferlenghi is certain that this discovery wouldn’t have been possible without the skills she learned at EMBL or the virus structures that she – and other researchers in the field – discovered. At EMBL, the overarching research aim is to advance human knowledge, but one benefit is that this can also improve human health. The question remains – who else’s blue-sky thinking could translate to the clinic, and when?
To learn more about vaccines and the viral diseases they protect against, watch talks given at EMBL’s Science and Society Conference, Infectious Diseases: Past, Present, and Future.


