Mothers and daughters
For the first time, scientists record how starfish egg cells eliminate crucial structures to ensure the embryo will be viable
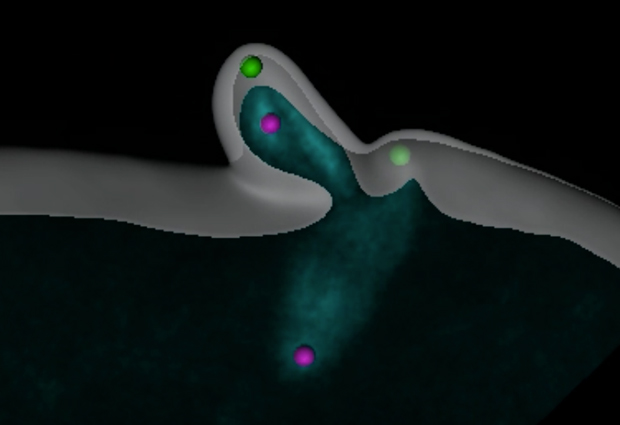
EMBL scientists have observed how an egg cell gets rid of its centrioles – structures that play a crucial role in cell division – to ensure the proper development of the embryo. The study, published today in Journal of Cell Biology, is the first time the whole process has been seen in its entirety, in real time.
The basics of sexual reproduction appear to be very simple: sperm plus egg cell equals embryo. But within cells, it gets trickier: simply combining the genetic content of two cells would lead to disaster; every generation would carry twice as much DNA as its parents. To prevent this, egg and sperm cells halve their genetic content before fusing. A similar issue arises with structures called centrioles.
People have tagged a mother centriole in the sperm, and found it still intact in the late embryo!
Centrioles act as anchors for the spindle apparatus, which pulls genetic material apart during cell division. If a fertilised egg has centrioles from both the egg cell and the sperm, its genetic material will be pulled in too many directions and it will be shared unevenly between the resulting cells, which is likely to make the embryo unviable. So in animals, before an egg cell is fertilised by a sperm, its centrioles are eliminated, ensuring that the resulting embryo receives only the sperm’s centrioles.
When a cell is dividing, each anchor point is actually a pair of centrioles: a mature ‘mother’ centriole, and an immature ‘daughter’ centriole.
“Mother centrioles are known to be very, very stable,” says Péter Lénárt from EMBL, who led the work. “In the worm C.elegans, people have tagged a mother centriole in the sperm, and found it still intact in the late embryo!”
To investigate how the egg cell manages to rid itself of such a resilient structure, Joana Pinto, a PhD student in the Lénárt lab, developed fluorescent tags for mother and daughter centrioles in a starfish egg cell and recorded the entire process of eliminating them.
She found that the egg cell expels the two mother centrioles (green), jettisoning them into the two ‘polar bodies’ that also serve as dumps for its surplus genetic material. One daughter centriole (purple) is also dragged into a polar body, leaving the other daughter centriole alone in the egg cell. “This only happens in egg cell formation,” says Lénárt. “In a normal division a single daughter centriole is never left alone.”
“It seems that if this daughter is alone it is unstable, and will be degraded,” says Pinto. “But if we make a mother centriole stay in the cell, it doesn’t get destroyed, so the fertilised egg ends up with a tripolar spindle and can’t divide.”
Thanks to further probing aided by an electron microscopy technique developed at EMBL by Yannick Schwab’s lab, the scientists found that mother centrioles are expelled into polar bodies thanks to little appendages that centrioles acquire as they mature. Their data suggests that these appendages direct mother centrioles to the cell membrane, ready for ejection.
The scientists would like to probe further into how mother centrioles are transported and ejected, and investigate how and why isolated daughter centrioles break down.
“Eggs are incredibly diverse – think of chickens, frogs, starfish – so I doubt that this is exactly the same in all animals,” says Lénárt. “But underlying that diversity are conserved modules like the centrioles. By understanding the molecular logic of how those modules can be combined in different ways, we can begin to reconstruct how this diversity evolved.”



