Targeted volume correlative light and electron microscopy of an environmental marine microorganism
Journal of Cell Science 09 August 2023
10.1242/jcs.261355
A new method developed by EMBL scientists can help us identify and investigate plankton species in field samples with greater speed, accuracy, and resolution than ever possible before
By Michelle Noble, Postdoctoral Fellow, Hentze Group
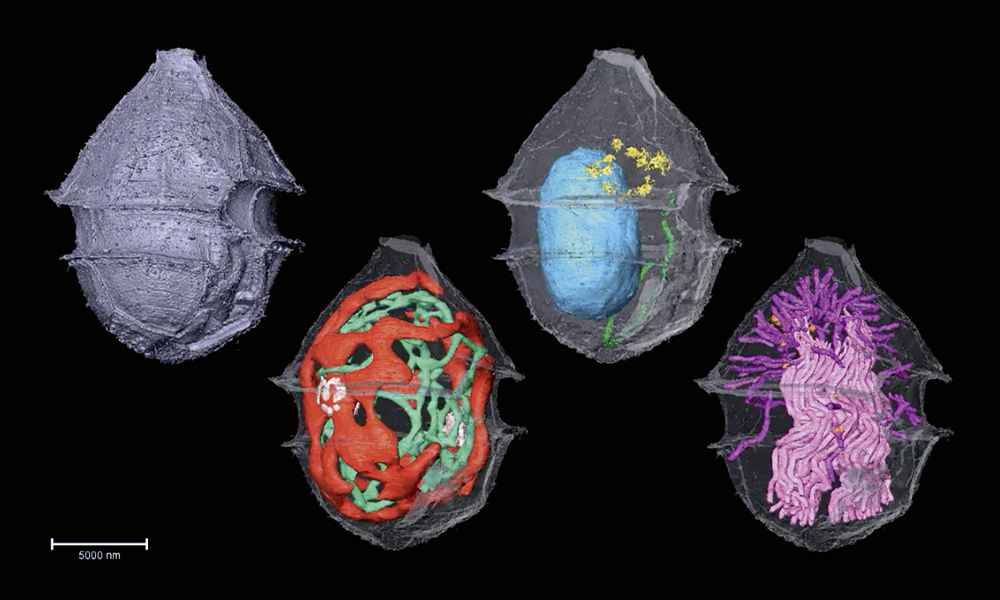
The image above shows a species of plankton – mostly unicellular organisms, billions of which drift in the world’s oceans. Photosynthesising plankton make a lot of the oxygen we breathe, fixing CO2 from the atmosphere in the process. Plankton also form the base of the ocean food pyramid, sustaining the complex ocean food webs. Too few of them, and life as we know it, would collapse. Too many plankton in some toxic ‘blooms’ can also be harmful, wreaking havoc on the ocean ecosystems. Given the life-sustaining weight these microscopic floaters carry, and the delicate balance of their population dynamics, it is surprising how little we know of them.
A research team led by Paolo Ronchi and Yannick Schwab from EMBL Heidelberg have recently developed a method that could help us accurately identify and study these organisms in the wild. By freezing the samples at high pressures near the ocean, and then using a combination of light and electron microscopy, they demonstrate how we can investigate plankton collected in the field in their natural state.
Oceanic samples typically contain hundreds of species, only a few of which survive in the laboratory. In the new method, after preparing the field samples in a way optimised for both light and electron microscopy, the researchers first shine light of different wavelengths on it to reveal fluorescing cells through confocal microscopy. This gives a broad overview of the diversity of cells in their collection.
Once they spot a cell of interest, researchers go in with more powerful, but time-consuming, tools like volume electron microscopy. This reveals the 3D makeup of the organism, allowing researchers to correctly identify the sample and make novel biological observations. This method, called targeted volume correlative light and electron microscopy (vCLEM), represents a major step forward in our ability to study the ultrastructure of plankton and other unicellular organisms.
In the image above, you can see the fine details of the phytoplankton Ensiculifera tyrrhenica revealed by this method, including their protective covering, called theca (metallic purple), energy-producing mitochondria (green), photosynthesising chloroplasts (red), nucleus (blue), protein sorting station called Golgi complex (yellow), secretory organelles called mucocysts (orange) and defensive organelles called trichocysts (pink and magenta).
The researchers plan to apply this technique to samples from the TREC expedition, which aims to document the biodiversity along the European coastline, as well as its interactions with the environment. “It is a promising method to make sense of the heterogeneous mixes that are collected in the wild,” said Paolo Ronchi, Scientist in EMBL’s Electron Microscopy Core Facility (EMCF).
The advanced mobile laboratory (AML) supporting the expedition is equipped with a high-pressure freezer and other cutting-edge technology that would help bring life along the coast into focus. “Capturing field samples in such a fresh state requires proximity between the machine and the sample. The AML enables this,” said Yannick Schwab, Team Leader and Head of EMCF.
Journal of Cell Science 09 August 2023
10.1242/jcs.261355



