
Picky eaters: how microglia capture their prey
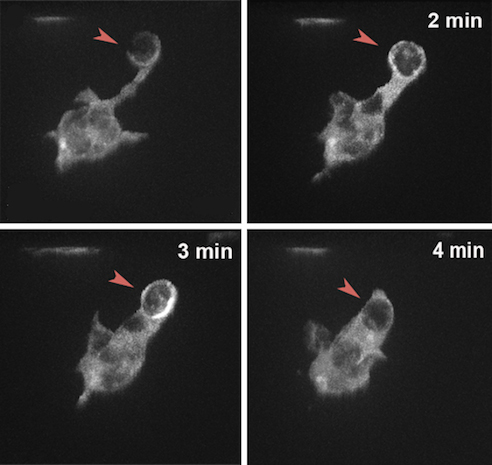
Dying cells are thought to produce an “eat me” signal that attracts immune cells, which destroy them by engulfing and digesting them. But it’s still unknown exactly how immune cells recognize that signal and find their targets, especially in the brain, where there is little space to move around. The brain contains specialized immune cells called microglia that work as housekeepers, identifying and clearing out dead and dying neurons. But rather than bustling around and bumping into healthy neurons, they sit in one place and send out thin tendrils to grab their targets and bring them back for digestion, like a diner at a banquet plucking one grape from a bowl of fruit halfway down the table.
Francesca Peri and her lab at EMBL Heidelberg recently investigated two proteins on the surface of microglia thought to be receptors that detect dying neurons: BAI1 and TIM-4. The team, along with Darren Gilmour and EMBL alumnus Jochen Wittbrodt, deactivated BAI1 and TIM-4 in living zebrafish embryos, first separately and then concurrently, to see what happened. Microglia without BAI1 had trouble forming phagosomes – the capsules that enclose and transport the neuron back to the cell for digestion. Those that lacked TIM-4 had trouble latching on to the neurons, often trying multiple times and failing, and their phagosomes tended to break open and spill their contents.
Microglia with non-functional BAI1 and TIM-4 had no problem finding their targets, they just couldn’t eat them properly, like a guest who eyes the roast beef hungrily but can’t get it into their mouth. “They have a fork, but somehow it doesn’t work,” says Peri. Faulty forks are especially problematic for microglia because they rely on their phagosomes to find and extract their target neurons, rather than moving to the cells and engulfing them directly. In the brain “what’s around you really matters – you can’t move around like an elephant,” Peri says.
If we can understand how these microglia go after neurons, we are learning something that has great value.
While her team’s study focused on how microglia behave during healthy development, Francesca thinks her research could potentially offer insight into neural disease as well. It is likely that microglia without functional phagosomes can’t prune neurons out of developing brains, and as a consequence crucial neural circuits may not form properly. A study published by fellow EMBL researcher Cornelius Gross in February 2014 found that mice with fewer microglia during development exhibit autistic-like behaviors throughout their lives, possibly because the connections between their neurons were not appropriately trimmed at a crucial stage. Other problems occur when microglia become too active and start digesting healthy neurons, like someone going into the kitchen and devouring the dessert before it’s put on the table. Francesca says that such overactive microglia could be a cause of degenerative diseases like multiple sclerosis and Alzheimer’s.
Microglia must be kept in check from both sides – either too much or too little activity can have severe consequences for an organism’s brain. Because removing neurons is so crucial for healthy brain function, there is likely some redundancy built into the process, with multiple molecules – and even cell types – able to perform the same duties in case one of them is out of commission. “We know that if we remove these microglia, other cells in the brain will eventually step in” and help clear dead neurons away, says Peri. That redundancy might mean that there is no one molecule that allows immune cells to recognize their targets, but rather a host of molecules that may perform that task at different times and in different situations. “We know that [microglia] are not sniffing around randomly – it’s a targeted migration [to their targets]. But we still don’t know which molecules work so precisely to direct each branch” to the dying neurons, she says.
Determining the roles of the many molecules that direct the processes of detecting neurons, capturing them in phagosomes and digesting them is a continuing research goal for her team. “If we can understand how exactly these microglia go after neurons and how exactly they are able to engulf and remove neurons, we are learning something that has great value.”
Watch a video of Peri’s microglia in action:


