Video Release: Filming life in the fast lane
New microscope takes a different angle – or four
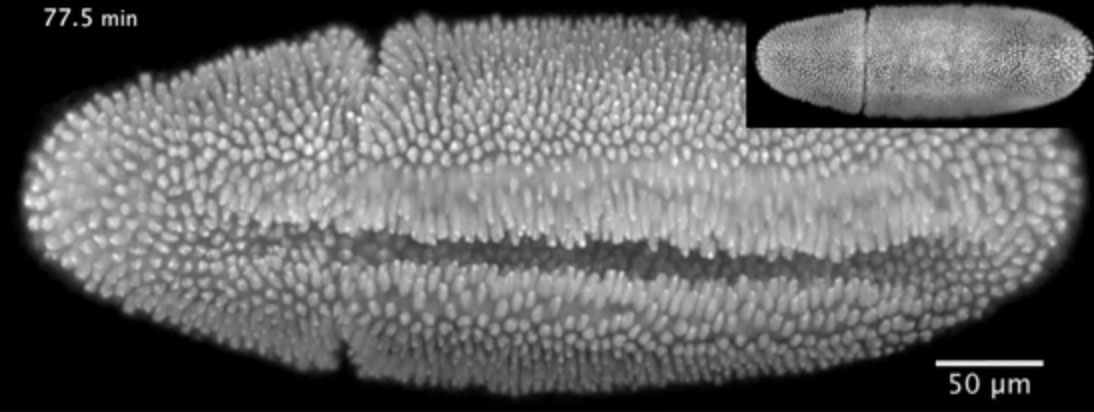
“This video shows a fruit fly embryo from when it was about two-and-a-half hours old until it walked away from the microscope as a larva, 20 hours later,” says Lars Hufnagel, from the European Molecular Biology Laboratory (EMBL) in Heidelberg, Germany. “It shows all the hallmarks of fruit fly embryonic development in three dimensions.”
The video was obtained using a new microscope created by Hufnagel’s lab and described online today in Nature Methods. In it, you can watch as cells on the embryo’s belly dive in to form what’s known as the ventral furrow. Other cells can then be seen moving around the embryo’s rear end to its back (inset), in a process called convergent extension. And later, when an opening appears in the embryo’s back (inset), see the surrounding cells close the gap in a process known as dorsal closure.
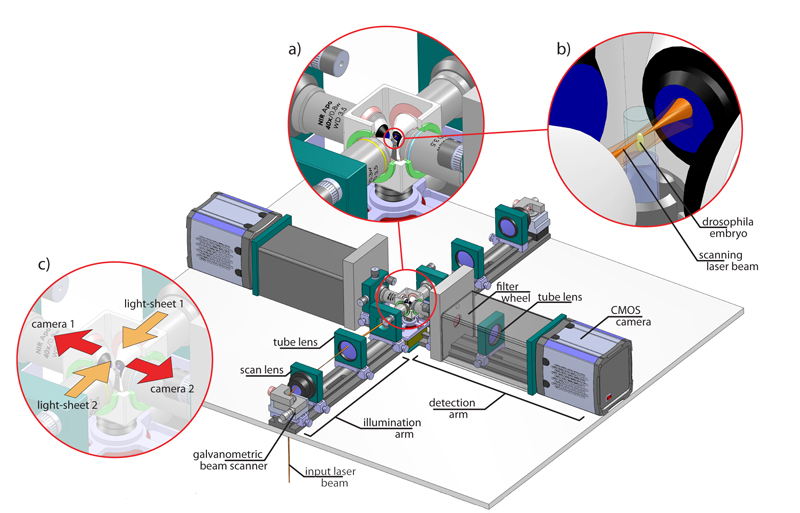
The new microscope, called Multi-View SPIM, or MuVi-SPIM for short, enables scientists to image rapid biological processes in thick samples at unprecedented detail. It builds upon the Selective-Plane Illumination Microscopy (SPIM) technology developed at EMBL a few years ago. Like SPIM, the new microscope shines a thin sheet of light on the embryo, illuminating one layer of a sample like this embryo at a time, to obtain an image of the whole sample with minimal light-induced damage. But MuVi-SPIM takes four full images from different angles, eliminating the need to rotate the sample. This speeds up the imaging and enables scientists to merge the four images into a single high-quality three-dimensional image for the first time.
Hufnagel and colleagues can obtain a high-quality three-dimensional image of a fruit fly embryo in a few seconds, and be ready to repeat the process. This means the different images that make up the video are taken in such rapid succession that very little has changed in the embryo from one frame to the next – so scientists can be sure of where each cell – or even structures inside cells – is and track it throughout the video. Using such fast imaging, Hufnagel and colleagues were able to record the movements of every nucleus in the embryo not only during the later developmental stages shown here, but also throughout the first three hours of the embryo’s life, when nuclei divide very rapidly.
“Everything in this microscope is optimised for speed and light-sensitivity,” says Uros Krzic from Hufnagel’s lab, who designed the microscope. “Not just the optics, mechanics and cameras, but also the software that controls the microscope and processes the data. We can finally get high-quality data in real time.”
In the future, the scientists would like to use their new microscope to investigate the how organs and tissues form, not only in the fruit fly but also in other model organisms.
The video is also available on the EMBL YouTube Channel: http://youtu.be/MefTPoeVQ3w.
Additional resources
Source article
Krzic, U., Gunther, S., Saunders, T.E., Streichan, S.J. & Hufnagel, L. Multi-view selective-plane illumination microscope for rapid 3D imaging of developmental processes with sub-cellular resolution. Nature Methods, Advance Online Publication 3 June 2012.
DOI: 10.1038/NMETH.2064.
Article abstract
We present a multi-view selective-plane illumination microscope (MuVi-SPIM),comprised of two illumination and two detection objective lenses, that allows rapid fluorescence imaging of large specimens over biologically relevant developmental time scales and with sub-cellular resolution. MuVi-SPIM records four high-resolution 3D images through four imaging branches in a few seconds. The fixed geometrical arrangement of the imaging branches eliminates the need for fiducial markers and computationally intensive data-driven post-processing methods, allowing registration and fusion of multi-view data into a single high-quality image in real-time. To demonstrate its potential, we image the extremely rapid early fly morphogenesis. We show that the high speed of MuVi-SPIM enables faithful tracking of nuclei in the blastoderm and in post-blastoderm tissues and investigation of cell shape changes during embryonic development.
Press Coverage
- NEW SCIENTIST TV , 6 JUNE 20123D movie captures fruit fly embryo turning into larva
- SCIENCES ET AVENIR (FRANCE) , 5 JUNE 2012[EN VIDEO] Le développement d’un embryon de mouche



