Constant overlap
EMBL scientists identify molecular machinery that maintains important feature of the spindle
During cell division, microtubules emanating from each of the spindle poles meet and overlap in the spindle’s midzone. Scientists at the European Molecular Biology Laboratory (EMBL) in Heidelberg, Germany, have uncovered the molecular mechanism that determines the extent of this overlap. In a study published today in Cell, they were able to reconstruct such anti-parallel microtubule overlaps in vitro, and identify two proteins which are sufficient to control the formation and size of this important spindle feature.
Thomas Surrey and his group at EMBL found that one protein, PRC1, bundles together microtubules coming from opposite ends of the cell, attaching them to each other. It then recruits a second protein, a molecular motor from the kinesin-4 subfamily, increasing its concentration in the spindle midzone. This motor walks along the overlapping microtubules like an officer on patrol, until it reaches one of the ends. When enough kinesin-4 molecules reach the end of the overlap, they inhibit the growth of microtubules there, thus keeping the overlap size constant without affecting microtubules elsewhere in the cell.
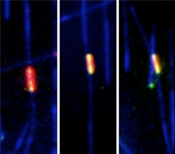
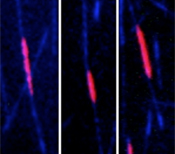
If both PRC1 and kinesin-4 are present (fist video), microtubules (blue) grow only until their overlap (red/yellow) reaches a certain size, which then remains constant. But if only PRC1 is present, microtubule growth is not inhibited in the overlap region, which becomes bigger and bigger (second video). Video credits: EMBL/P. Bieling
The spindle midzone plays an important role not only in helping to align the chromosomes in metaphase, but also in the final stages of cell division, when it drives the physical separation of the two daughter-cells. But between these two stages, the two ends of the spindle must move away from each other, to drag half the genetic material to each side of the dividing cell. At this point, if PRC1 and kinesin-4 had stopped microtubule growth permanently in the central part of the spindle, the overlap would become smaller and smaller, until eventually the spindle itself would collapse, jeopardising cell division. But Surrey and colleagues found that PRC1 and kinesin-4 control the overlap size in an adaptive manner. As the spindle stretches and the overlap between microtubules becomes smaller, the scientists posit, the inhibitory effect of kinesin-4 diminishes, allowing the microtubule ends to grow.
“Our findings show how molecules millionths of millimetres small can control the size of a structure about a thousand times larger than themselves,” Surrey concludes: “they help us to understand the fundamentals of cell division.”