Poster prizes at ‘Molecular mechanisms in evolution and ecology’
Our EMBO Workshop ‘Molecular mechanisms in evolution and ecology’ is a long-standing conference series, which has become known for exploring yeasts as models to study questions of general relevance to evolution and ecology. It covers a range of topics, including evolution theory, ecology, genetics, cell biology, genomics, systems biology, metabolic modelling and engineering. We were happy to hold this meeting in person again, with 125 people attending on site (and 30 virtually). It was a fantastic opportunity to meet for a four days’ programme full of exciting science, exchanging ideas, presenting latest research, catching up with old friends and making new ones. There were 66 posters presented up on the helices of the Advanced Training Centre, with two poster sessions during which the presenters could discuss their research – their work was then voted for by other attendees and speakers. We are pleased to share with you the four best poster prize winners plus the additional prize for the best-timed flash talk!
Best poster prizes:
The benefit of being slow – a long lag phase yields a fitness advantage in constant environments
Presenter: Lloyd Cool

Abstract:
Microorganisms are often confronted with changes in nutrient availability. Adaptation to these fluctuations takes time and energy, which results in a period of reduced or no growth; called lag. Interestingly, genetically distinct Saccharomyces cerevisiae strains often show vast differences in lag duration for the same shift in carbon source. Furthermore, even within isogenic populations, individual cells respond heterogeneously to this change. This variation in lag duration between different strains and individual cells raises that question whether a long lag might be favorable in certain cases.
A previous QTL analysis from our lab indicates that natural sequence variation in the gene YLR108C is at the basis of variation in lag duration between different S. cerevisiae strains. This observation allows us to specifically study the trade offs and fitness benefits of different lag durations by using strains having the same genetic background but only differing in YLR108C alleles. Competition experiments between these strains show that strains with a short lag have a fitness advantage in fluctuating carbon environments, while the YLR108C allele known to cause a long lag in fluctuating carbon environments confers the highest fitness in environments with a stable glucose concentration. Interestingly, natural variation in YLR108C across 1,011 natural S. cerevisiae strains suggests a link between specific SNPs and the ecological niche from which the strains carrying these alleles were isolated. Specifically, strains isolated from niches that contain mixed carbon sources often contain an allele of YLR108C that yields a short lag phase, while strains found in niches that typically contain high levels of preferred carbon sources like glucose often carry a YLR108C allele associated with a long lag.
Taken together, our results suggest that variation in YLR108C and hence the variation in lag duration could be evolutionarily tuned to specific ecological niches and the glucose supply in the environment.
Yeast adaptive evolution towards the cellulosic ethanol
Presenter: Jeferson Gross
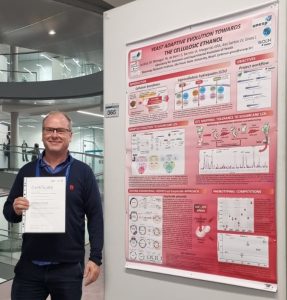
Abstract:
Saccharomyces cerevisiae fermentation of biomass-derived sugars for ethanol production depends on two technical innovations: (1) the establishment of metabolic pathways for xylose consumption, a C5 sugar rich in lignocellulosic hydrolysates (LCHs); (2) and the development of robust yeasts capable of tolerating lignocellulosic inhibitors (weak acids, phenols, and furfurals) generated upon biomass hydrolysis. In a vigorous research program involving metabolic engineering, QTL mapping, and adaptive laboratory evolution (ALE), we are developing strains suitable for production of cellulosic ethanol. An innovative QTL mapping approach, based reiterated rounds of selection and backcrossing, allowed us to chart in the Brazilian bioethanol strain PE-2_H4 adaptive alleles for LCH tolerance related to the genes VPS70, MKT1, HAP1, PHO84, FLO8 and CRF1. Allele swapping of QTL into the inferior parental conferred LCH tolerance to the sensitive S288C lab strain. Through three ALE experiments with 8 replicate populations of strains CEN.PK113-7D and PE-2_H4 we identified, via WGS after 550-750 generations, about 70 alleles conferring tolerance to sugarcane LCHs. Common adaptation pathways involve protein SUMOylation/ubiquitination (SIZ1), chromatin modification (CHD1), rRNA synthesis (RET1), and protein stability (SSB2). The highest degree of evolutionary parallelism is found for alleles putatively downregulating the functions of the plasma membrane H+-ATPase (PMA1 and HRK1), suggesting that energy conservation (i.e., high ATP/ADP rations) might be a major driving force for LCH tolerance. A different metabolic and evolutionary engineering approach resulted in yeasts with 12-18 copies of heterologous expressed xylose isomerase and widespread mutations in genes regulating the iron-sulfur cluster homeostasis. A xylose-consuming yeast population has been crossed with a pool of LCH-tolerant ALE strains to generate robust yeasts selected for efficient production of cellulosic ethanol.
Bacterial fungal co infections and their role in the evolution of antimicrobial resistance
Presenter: Lisa Juliane Kahl

Abstract:
Microorganisms routinely exist in multispecies communities that exchange metabolites, sense each other’s presence, and exchange genetic material, including resistance genes. Clinical data show that most patients are simultaneously infected by multiple species. Despite growing evidence that infections that involve multispecies communities are i) more difficult to treat and ii) a source of antimicrobial resistance, microbial pathogens have so far typically been studied in isolation. Hence, we have focused on a clinically relevant co infection community by studying the interactions between a problematic bacterial (Pseudomonas aeruginosa) and a fungal (Candida albicans) pathogen. The multi drug resistant bacterium Pseudomonas aeruginosa (PA) is frequently co isolated with the opportunistic fungal pathogen Candida albicans (CA) and increased antibiotic tolerance of PA has been observed in dual species biofilms with CA. In addition, both pathogens are known to occupy multiple infection niches in the human body, including the lungs and urinary tract.Here, we have established a co infection community model and probed how the proteomic make up of both PA and CA shifts in comparison to single species growth. This experiment revealed that expression of biochemical warfare, but also general metabolic processes are modulated in both species upon co culturing. In addition, vast metabolic adaptations become apparent when culturing these pathogens in different metabolite environments that mimic lung and urinary tract infection sites. We show that these changes shape the pathogens’ response to various antimicrobial agents and may lead to altered evolution dynamics of the individual species upon drug stress.Future work will aim to identify the key mechanisms that PA and CA use to evade antimicrobial treatment and investigate how these interactions contribute to the emergence of tolerant and resistant phenotypes within a host. These findings will inform the development of novel drugs and treatment strategies to combat this emergent health threat.
Due to the confidentiality of the unpublished data, we cannot share the poster.
Population structure of S. cerevisiae from agave fermentations in Mexico
Presenter: Lucía Morales
Abstract:
Agave spirits are traditional beverages of remarkable importance in Mexico. They are produced through the fermentation of cooked agave hearts by yeasts and bacteria living between the factories and the variety of surrounding ecosystems that characterize this megadiverse country. In this work, we collected S. cerevisiae isolates from fermenting agave juice in 62 different locations in Mexico, and analyzed their evolutionary relationship, genetic diversity, and population structure. The genomes of 169 isolates revealed a correlation between the strains´ isolation site and their genetic relatedness. Interestingly, we found two major clusters of only Mexican strains. We also checked the agave strains for admixture signals in their genomes to analyze if the strains have been genetically isolated from one another. Our findings show we have identified a group of S. cerevisiae isolates that are genetically different from strains found in other regions of the world with high levels of introgression from the sister species S. paradoxus.
Best-timed flash talk:
Autotrophic adaptive laboratory evolution of the acetogen Clostridium autoethanogenum delivers a gas fermenting strain with superior growth, products, and robustness
Presenter: Henri Ingelman

Abstract:
Microbes able to recycle gaseous one carbon (C1) feedstocks including CO2 are of growing importance in transitioning to sustainable production chemicals and fuels and a circular economy. Acetogens are particularly interesting biocatalysts since gas fermentation using Clostridium autoethanogenum has already been commercialized. Most non commercial acetogen strains, however, display slow growth and poor product profiles and require complex media. In this work, we adapted the wild type acetogen C. autoethanogenum in three different lineages on CO or syngas (CO+CO2+H2) for ability to grow on minimal medium and for faster growth using a combination of serial transfers in bottles, chemical mutagenesis, and chemostat evolution. After significant improvement in the population maximum specific growth rates (μmax) on minimal medium, single colonies were isolated and studied in autotrophic bottle batch and bioreactor chemostat cultures. Evolved isolates exhibited superior product patterns in all studied conditions and ~2 3 fold higher μmax than the wild type starting strain. Notably, one evolved strain S232868 exhibited supreme performance in terms of μmax, product profile, and robustness in operating continuous bioreactor cultures. Next, whole genome sequencing of selected isolates was performed to identify the causal mutations related to improved phenotypes. This revealed certain hot spots in the genome of C. autoethanogenum under stronger evolutionary pressure. Evolved traits could be further explained on genotype phenotype level by combining bioreactor off gas analysis with multi omics analysis and genome scale metabolic modelling. The detected mutations serve as useful candidate sites and proteins for further studies and metabolic engineering of superior cell factories. Additionally, this work provides a robust C. autoethanogenum base strain, S232868, to facilitate generation of higher quality datasets through the use of minimal medium and bioreactor chemostat cultures.
Due to the confidentiality of the unpublished data, we cannot share the poster.
The EMBO Workshop ‘Molecular mechanisms in evolution and ecology’ took place from 5 – 8 October 2022 at EMBL Heidelberg and virtually.