Havoc in biology’s most-used human cell line
Genome of HeLa cells sequenced for the first time
In a nutshell:
– Scientists deliver the first high-resolution sequence of HeLa cells, a key research tool for human disease and general biology
– Sequence analysis reveals the full extent to which HeLa cells are different to the Human Genome Project reference
– Resource could enhance the quality of research using HeLa cells
HeLa cells are the world’s most commonly used human cell lines, and have served as a standard for understanding many fundamental biological processes. In a study published today in G3: Genes, Genomes and Genetics online, scientists at the European Molecular Biology Laboratory in Heidelberg, announce they have successfully sequenced the genome of a HeLa cell line. It provides a high-resolution genomic reference that reveals the striking differences between the HeLa genome and that of normal human cells. The study could improve the way HeLa cells are used to model human biology.
The scientists’ analysis of the HeLa genome revealed widespread abnormalities in both the number and structure of chromosomes, as well as factors commonly associated with cancer cells like losing healthy copies of genes. In particular, the researchers found that countless regions of the chromosomes in each cell were arranged in the wrong order and had extra or fewer copies of genes. This is a telltale sign of chromosome shattering, a recently discovered phenomenon associated with 2-3% of all cancers. Knowledge of the genetic landscape of these cells can inform the design of future studies using HeLa cells, and strengthen the biological conclusions that can be made from them.
“The results provide the first detailed sequence of a HeLa genome,” explain Jonathan Landry and Paul Pyl from EMBL, who carried out the research. “It demonstrates how genetically complex HeLa is compared to normal human tissue. Yet, possibly because of this complexity, no one had systematically sequenced the genome, until now.”
“Our study underscores the importance of accounting for the abnormal characteristics of HeLa cells in experimental design and analysis, and has the potential to refine the use of HeLa cells as a model of human biology,” adds Lars Steinmetz from EMBL, who led the project.
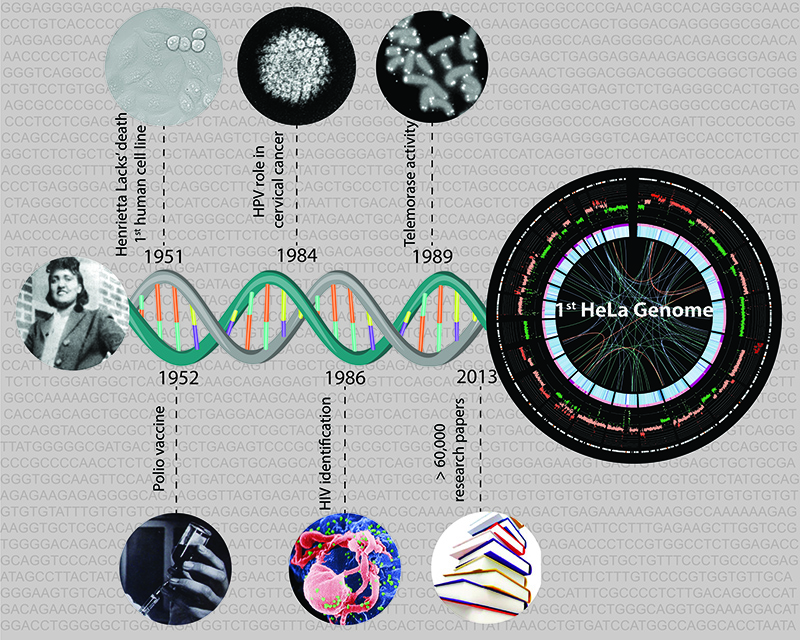
For decades HeLa cells have provided effective and easily usable biological models for researching human biology and disease. They are widely regarded as the ‘industry standard’ tool for studying human biology. Studies using them have led to two Nobel prizes and a host of advancements in many areas, including cancer, HIV/AIDS and the development of the polio vaccine. The HeLa genome had never been sequenced before, and modern molecular genetic studies using HeLa cells are typically designed and analysed using the Human Genome Project reference. This, however, misrepresents the sequence chaos that characterises HeLa cells, since they were derived from a cervical tumour and have since been adapting in laboratories for decades.
The study provides a high-resolution genetic picture of a key research tool for human biology. It highlights the extensive differences that cell lines can have from the human reference, indicating that such characterisation is important for all research involving cell lines and could improve the insights they deliver into human biology.
We updated this Q&A section as the original one contained an unfortunate factual error, which as many scientists have noticed, was contradictory to the information given in the article. This happened as the result of lack of adequate supervision of the preparation of the Q&A section. EMBL is implementing means to ensure that similar lapses do not occur in future.
HeLa cells – a guide
What are HeLa cells?
HeLa cells originate from the cervical cancer tumour of a patient named Henrietta Lacks (hence their name), who later died of her cancer in 1951. They were the first type of human cells that were successfully cultivated in the laboratory, and have since been the most widely used human cell line in biology. Cancer cells by definition continuously replicate themselves, thus making good candidates for experimental biology; HeLa cells were particularly aggressive at doing so and thus became very popular. There are lots of different subtypes of HeLa cells that are now used – the study by EMBL researchers has sequenced the genome of one subtype of HeLa cell.
How have HeLa cells been used in science?
The use of HeLa cells has contributed to scientists’ understanding of a wide range of fundamental biological processes and more than 60,000 research publications. One of the earliest uses of HeLa cells was to develop the vaccine against the polio virus. Recently, two Nobel prizes have been awarded for discoveries where HeLa cells played a central role: the link between human papillomavirus and cervical cancer (2008, Harald zur Hausen) and the role of telomerase in preventing chromosome degradation (2011, Elizabeth Blackburn, Carol Greider, and Jack Szostak). During the past decade, HeLa cells have been used to develop large-scale technologies like RNA sequencing, RNA interference screens, and other ‘omics’ approaches. Studies using these approaches on HeLa cells have furthered our understanding of important cellular processes, such as cell division. HeLa cells have also been used in studies of nuclear toxins, and have even been into space.
Why are HeLa cells so useful?
Biologists tend to use HeLa cells as the ‘default’ human cell line because they are so easy to work with – they reproduce rapidly, easily and cheaply. Despite being cancerous, HeLa cells still share many basic characteristics with normal cells – they produce proteins, express and regulate genes, communicate with one another, and are susceptible to infections. It is thus possible for scientists to use HeLa cells to study not only cancer, but also basic functions carried out by all human cells. The genome sequence should make genetic studies using HeLa even more effective.
Can we infer anything new about Henrietta Lacks or her descendants from this sequencing?
The HeLa cell line sequenced in this study has spent decades in labs, dividing and thus undergoing mutations and changes – it is very different from the original cells that started growing in 1951. Furthermore, the original cells were taken from Mrs. Lacks’ cervical cancer – as cancer is a disease of the genome, the DNA of cancer cells is different to that of the patient. Therefore, the genome we sequenced contains a combination of genetic variants originating from the donor’s genome, variants that arose during the tumour’s development, and variants that occurred during the many years of in-lab evolution of the cell line. Comparisons to common genetic variants in human populations today allow one to infer variants likely to have been present in the donor’s genome, although such inferences can only be made with a certain likelihood. Measurements of common variants in the HeLa genome have been published before this study (e.g. SNP arrays) and further variants can also be inferred from HeLa DNA sequencing data already deposited in the public domain (e.g. ChIP-Seq and RNA-Seq). The data generated in this study therefore does not change the fact that it is possible to make predictions about Mrs. Lacks’ genome, or those of her descendants. The goal of this study is not to gain insights into Mrs. Lacks’ personal biology, but rather to provide a resource for researchers using HeLa cells. The value of our study is in creating a comprehensive resource of all variants (common and non-common) present in a cell line that has been of such high value for biomedical research.



