From foe to friend: mosquitoes that transmit malaria may help fight the disease
EMBL scientists identify gene behind malaria-resistant mosquitoes
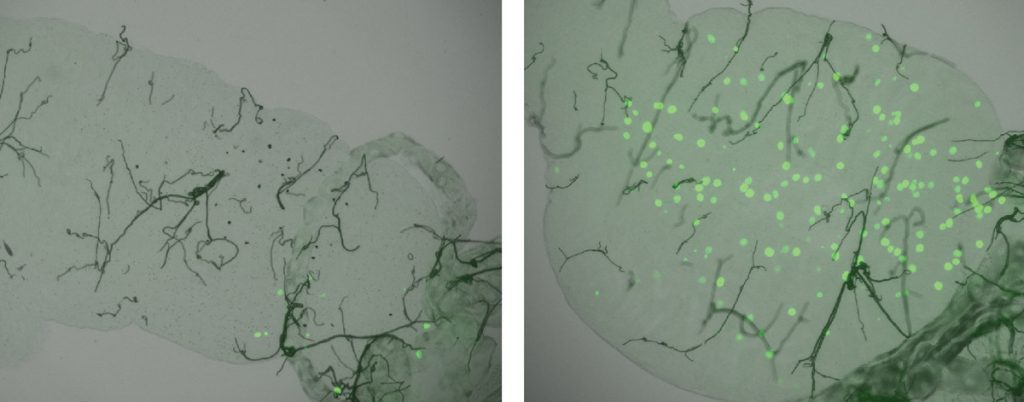
Click on image to download a high resolution version (tiff).
For many years, the mosquitoes that transmit malaria to humans were seen as public enemies, and campaigns to eradicate the disease focused on eliminating the mosquitoes. But, as a study published today in Science shows, the mosquitoes can also be our allies in the fight against this common foe, which kills almost one million people a year and heavily impairs the economies of affected countries. In this study, researchers at the European Molecular Biology Laboratory (EMBL) in Heidelberg, Germany, and the Institut National de la Santé et de la Recherche Médicale (INSERM) in Strasbourg, France, discovered that variations in a single gene affect mosquitoes’ ability to resist infection by the malaria parasite.
“Malaria parasites must spend part of their lives inside mosquitoes and another part inside humans, so by learning how mosquitoes resist malaria, we may find new tools for controlling its transmission to humans in endemic areas”, says Stephanie Blandin from INSERM, who carried out the research at EMBL in collaboration with Lars Steinmetz’s group and with Rui Wang-Sattler (now at the Helmholtz Zentrum in Munich, Germany).
The scientists looked for clues in the genome – the whole DNA – of Anopheles gambiae mosquitoes, a major carrier of the parasite that causes the most severe form of human malaria in Africa. They focused on the mosquitoes’ resistance to a commonly used model organism: Plasmodium berghei, a parasite that causes malaria in rodents.
When they compared the genomes of mosquitoes that could resist this infection to those of mosquitoes that couldn’t, the scientists discovered that the major difference lies in a single section of one chromosome. Of the roughly 975 genes contained in this section of DNA, one in particular appears to play an important role in determining a mosquito’s resistance to malaria. This gene, called TEP1, encodes a protein which was known to bind to and promote the killing of Plasmodium berghei malaria parasites in the mosquito’s midgut, and the scientists discovered that their strain of resistant mosquitoes had a form, or allele, of TEP1, that was different from those found in non-resistant (or susceptible) strains.
To investigate whether this difference in alleles caused the variation in the mosquitoes’ resistance to malaria, the scientists developed a new technique, reciprocal allele-specific RNA interference, inspired by one Steinmetz’s group had previously created to study yeast. “This was a breakthrough, because the new technique is applicable to many different organisms”, says Steinmetz. “It extends the power we gained in yeast: we can go from a whole region of DNA to the actual causative gene – a feat rarely achievable in complex organisms”. The technique enables scientists to identify exactly which allele is behind a specific trait. They produced individual mosquitoes that had one TEP1 allele from the resistant strain and another from a susceptible strain, and then “turned off” – or silenced – one or other of these alleles. The result: silencing different alleles produced mosquitoes with different degrees of resistance to malaria, meaning that an individual mosquito’s resistance to the malaria parasite depends largely on which form(s) of this one gene it carries.
Although this study focused on the parasite that causes malaria in rodents, there is evidence that this gene may also be involved in the mosquitoes’ immune response to human malaria – a connection the scientists are exploring, and which they believe may help to make malaria eradication programs more effective.



