
From cell to mouse – how tissues differentiate
Two new studies shed light on the mechanisms that control the fate of embryonic cells.
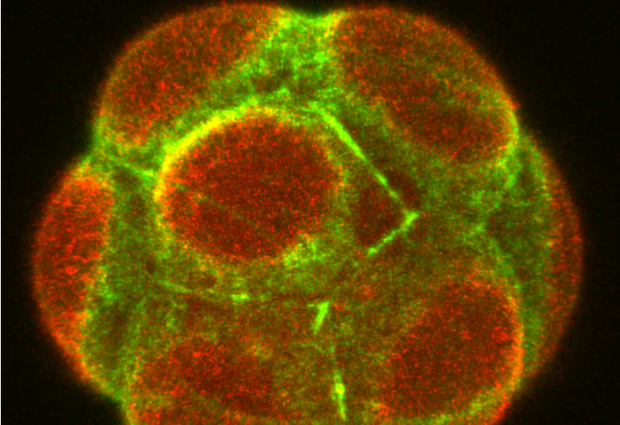
At a glance, the first cells of an embryo all seem identical. Yet, as they divide, they arrange into different tissues and follow a pattern that is common to many organisms. Mammalian embryos, before they adhere to the wall of the uterus, consist of a sphere with an outer layer of cells – the trophectoderm – that enfolds the inner cell mass, made of the epiblast and the primitive endoderm.
The way cells orient as they divide is crucial for the development of these tissues, as it modulates the position and fate of each cell. Just as the Earth has two poles with distinct characteristics, many cells are polar: they have an apex (pointing to the outside of the embryo) and a base (pointing inwards). In some cases, a cell divides symmetrically, meaning the cell contents are split equally between the two daughter cells, with each one sharing part of the apex and part of the base. Alternatively, it can divide asymmetrically along the ‘equator’, with the cell contents shared unequally between the daughter cells.
What controls how cell division goes is, in part, cell geometry and polarity. A sphere-like cell will tend to split asymmetrically, while elongated cells, stretched along their equator, will divide symmetrically. However, the situation is rarely so clear cut, and cells may have an intermediate shape. Ritsuya Niwayama and his colleagues in the Hiiragi Group at EMBL realised there must be a mechanism to prevent embryos from straying from the optimal developmental path.
In a new study published in Developmental Cell, they monitored cell division, volume, and shape changes in mouse embryos using live imaging with a light-sheet microscope. Niwayama and his group discovered that cell shape and polarity compete in a tug-of-war to determine how cells divide – symmetrically or asymmetrically.
The good news is that the interplay between cell shape and polarity allows embryos to be more resilient to disturbance. The researchers experimentally altered the symmetry of mouse embryonic cells and discovered that they show plasticity in dividing one way or the other until the disturbance is removed and normal development can resume.
This fail-safe mechanism ensures the right proportions of cells are allocated to the trophectoderm and to the inner cell mass in the embryo. While the inner cell mass further differentiates into the epiblast and primitive endoderm, a fluid-filled pocket (the lumen) forms and expands in the embryo. The Hiiragi Group investigated the interaction between the lumen and surrounding tissues in a second study led by Allyson Ryan, also published in Developmental Cell.
The researchers experimentally prevented lumen expansion to see whether it impaired the differentiation of the inner cell mass. The epiblast and primitive endoderm did not form or position properly, revealing the importance of lumen expansion for development. However, injecting a signalling protein known to specify the fate of the primitive endoderm, FGF4 (Fibroblast Growth Factor 4), partially rescued these abnormal embryos.
When Ryan and her colleagues inhibited the action of this protein in embryos bearing a normal lumen, they too failed to keep growing normally, highlighting the crucial role of FGF4 for inner cell mass differentiation. Thus, mechanical and chemical cues from the lumen are integrated to guide tissues to differentiate.
This research provides new insight into the development of early mammalian tissues. While many things can go wrong, evolution has provided embryos with sufficient plasticity to try to overcome the vagaries of life. The next step for this field of research will be to determine the precise molecular mechanisms behind the orientation of cell division and the specification of different embryonic tissues.


